Review
HJOG 2026, 25 (2), 121-134| doi: 10.33574/hjog.0622
Elizabet C. Jusuf1, Karloman A. Paipinan2
1Hasanuddin University Department, Makassar, Indonesia
2Kombeng Primary Health Care Center, Kutai Timur, Indonesia
Correspondence: Elizabet Catherine Jusuf, e-mail: elisabetjusuf@gmail.com
Abstract
Background: Nuchal translucency (NT) measurement is widely used in prenatal screening to detect chromosomal abnormalities, but its role in identifying congenital defect remains variably reported. Congenital heart disease (CHD) refers to structural malformations of the heart and/or great vessels, which predispose affected individuals to multiple morbidities across their lifespan and contribute to reduced long-term survival.. Although numerous studies have evaluated NT, none have specifically summarized the diagnostic sensitivity and specificity that reflect how well this screening performs when applied independently without adjunctive examinations. To address this gap, this systematic review was conducted to synthesize findings from previous studies and provide evidence-based insights into the diagnostic performance of NT for early detection of CHD.
Methods: A systematic review was conducted following PRISMA guidelines. PubMed, Embase, and Web of Science were searched from inception to August 2025 for studies assessing NT measurement in detecting CHDs and other structural anomalies. Eligible studies included prospective or retrospective designs involving pregnant women, with NT assessed at any gestational stage and outcomes confirmed by postnatal examination, autopsy, or detailed follow-up imaging. Data extraction included study characteristics, NT cut-off values, diagnostic accuracy metrics, and risk of bias. Due to heterogeneity in methods and outcomes, findings were synthesized qualitatively. This study was registered prior to the study in PROSPERO CRD420251125707.
Results: Twenty studies published between 1999 and 2023 met the inclusion criteria, encompassing diverse populations from Asia, Europe, South America, the Middle East, and Africa. NT cut-off values and measurement protocols varied widely. For CHDs, reported sensitivities ranged from 0.15 to 1.00 and specificities from 0.62 to 0.98, with most studies showing high specificity but variable sensitivity. NT measurement demonstrated higher accuracy for certain complex cardiac anomalies but lower performance for minor defects. For other structural anomalies—including neural tube defects, abdominal wall defects, and skeletal dysplasias—NT measurement showed moderate to high specificity but variable sensitivity, and performance was enhanced when combined with other ultrasound markers or biochemical screening. Risk of bias was generally low to moderate, with variability mainly due to study design and follow-up completeness.
Conclusion: NT measurement is a valuable early screening tool for CHDs and other major structural anomalies, particularly when used alongside targeted follow-up imaging. Its high specificity supports its use in risk stratification, but variable sensitivity underscores the need for standardized protocols and integration with complementary screening methods.
Keywords: Nuchal translucency, congenital heart defects, anomalies, prenatal screening, diagnostic accuracy
Introduction
Congenital heart disease (CHD) refers to structural malformations of the heart and/or great vessels, which predispose affected individuals to multiple morbidities across their lifespan and contribute to reduced long-term survival. CHD are the most common congenital malformations, affecting approximately 8–12 per 1,000 live births, and are a leading cause of perinatal morbidity and mortality (1). Early detection of CHD allows for timely referral to tertiary care, prenatal counseling, and delivery planning, which can significantly improve neonatal outcomes (2). First-trimester screening has traditionally focused on detecting chromosomal abnormalities, but advances in ultrasound technology have broadened its role to include structural anomaly assessment (3). Among the markers assessed during first-trimester ultrasound, nuchal translucency (NT) measurement-performed between 10+0 and 13+6 weeks gestation has gained recognition not only for its association with aneuploidy but also for its potential role in detecting major structural anomalies, including CHD (4).
Several studies have demonstrated a correlation between increased NT thickness and a higher risk of CHD and other major fetal malformations, even in karyotypically normal fetuses (5). The proposed mechanisms include cardiac dysfunction, altered lymphatic drainage, and developmental abnormalities that manifest early in gestation (6). NT measurement is a relatively simple, non-invasive marker that can be incorporated into routine first-trimester scans, making it an attractive tool for early risk stratification (7). However, its predictive value for CHD and non-cardiac structural anomalies varies across studies, influenced by factors such as NT threshold, population characteristics, sonographer expertise, and the use of additional ultrasound markers or biochemical tests (8, 9).
Despite an increasing number of studies exploring NT’s role in structural anomaly detection, the evidence remains fragmented, with inconsistent methodologies, heterogeneous populations, and varying definitions of abnormal NT. To date, no comprehensive synthesis has evaluated the diagnostic accuracy of first-trimester NT measurement specifically for detecting CHD and other major structural anomalies in a broad clinical context. Therefore, this systematic review aims to collate and critically appraise existing evidence, quantify diagnostic performance where possible, and identify factors influencing accuracy, with the goal of informing clinical practice and guiding future research.
Methods
Study design
This study was conducted as a systematic review following the updated guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) (10). The protocol and methodology were developed to ensure transparency, replicability, and methodological rigor in appraising evidence on the diagnostic accuracy of NT measurement for the early detection of congenital heart defects and other major structural anomalies.
Literature search strategy
A comprehensive literature search was conducted in PubMed, Embase, and Web of Science from database February to August 2025. Both controlled vocabulary (e.g., MeSH and Emtree terms) and free-text keywords related to NT measurement, congenital heart defects, and structural anomalies were employed. Search terms included: “nuchal translucency,” “first trimester screening,” “congenital heart defect*,” “CHD,” “structural anom*,” “major congenital abnormalit*,” “prenatal ultrasonography,” and related synonyms. Boolean operators (AND, OR) and truncation were applied as appropriate. Search strategies were adapted to the syntax and subject headings of each database. The search was restricted to human studies and English-language publications. Reference lists of all included studies and relevant reviews were screened manually to identify additional eligible records. The complete search strategies for each database are presented in Table 1.
Eligibility criteria
Studies were eligible if they involved pregnant individuals undergoing nuchal translucency measurement, with subsequent confirmation of congenital heart defects and/or other major structural anomalies through prenatal detailed echocardiography and/or postnatal examination. Eligible designs included randomised controlled trials, cohort studies, cross-sectional studies, and case–control studies reporting diagnostic accuracy measures (e.g., sensitivity, specificity, predictive values) or providing sufficient data to construct 2×2 tables. Studies were excluded if they focused on second-trimester nuchal fold rather than NT, reported only chromosomal outcomes without separate structural anomaly data, or were case reports, narrative reviews, editorials, in vitro or animal studies, or conference abstracts lacking full text.
Study selection and data extraction
All search results were imported into a reference management tool, and duplicate records were removed. Two reviewers independently screened titles and abstracts, followed by full-text assessment against the eligibility criteria. Disagreements were resolved through discussion or consultation with a third reviewer. Data extraction was performed using Endnote to capture: study design, year, country, sample size, participant characteristics, NT measurement protocol, reference standard used, type of anomalies assessed, NT cut-off values, diagnostic accuracy metrics, and follow-up details. Detailed quality assessment results are presented in Table 2.
Data synthesis
Due to heterogeneity among studies in terms of NT cut-off values, measurement protocols, study designs, population risk profiles, and outcome assessment methods, quantitative pooling of results was not always feasible. Therefore, a qualitative descriptive synthesis was conducted. Findings related to the diagnostic accuracy of NT measurement for detecting congenital heart defects and other structural anomalies were summarised narratively to evaluate its effectiveness in early detection compared with other screening approaches or standard care.
Result
Study characteristics
The initial database search yielded 894 records, of which 594 remained after duplicate removal (Figure 1). Following screening of titles and abstracts, 36 full-text articles were assessed for eligibility. Ultimately, 20 studies met the inclusion criteria and were incorporated into this systematic review. These studies were published between 1999 and 2023, representing a cumulative sample of over 20 pregnancies assessed for nuchal translucency in relation to congenital heart defects and other major structural anomalies.
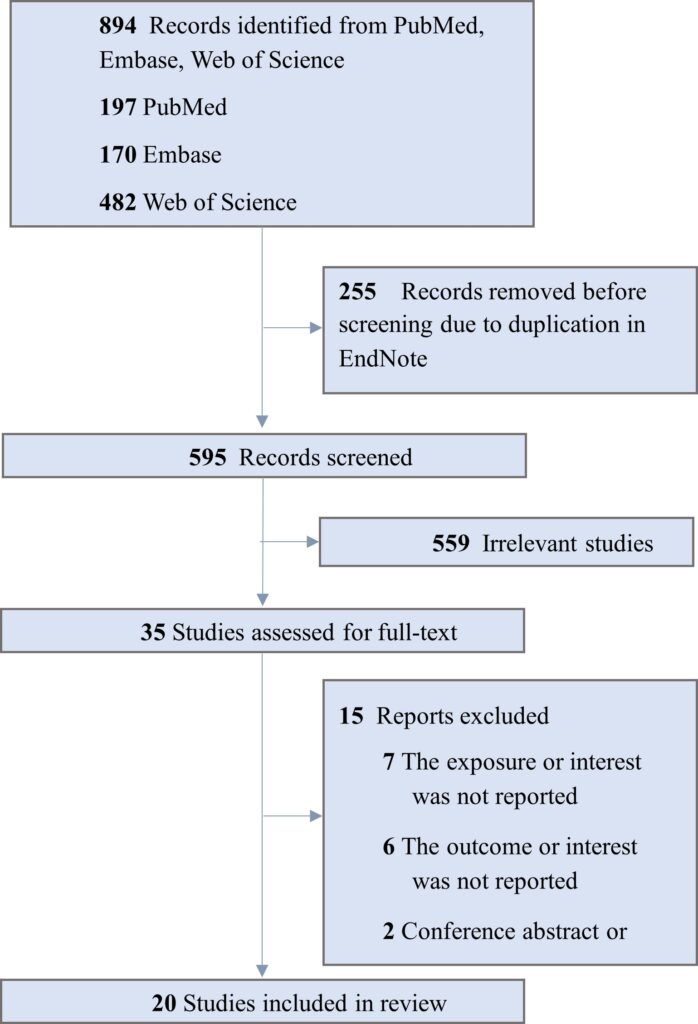
Figure 1. ROC Curve for PT in Prediction of Macrosomia.
The studies were conducted across multiple regions, including Asia, Europe, South America, and Africa, with both single-center and multicenter designs. Sample sizes ranged from fewer than 100 participants to over 10,000, and gestational age at NT assessment varied from early first trimester to late second trimester, depending on the study objectives. Most studies used standardized ultrasound protocols to measure NT, but definitions of increased NT differed, employing either fixed cut-offs (e.g., ≥3.0 mm or ≥3.5 mm) or percentile-based thresholds (≥95th or ≥99th percentile).
Diagnostic confirmation methods included detailed fetal anomaly scans, targeted echocardiography, genetic testing where indicated, and postnatal follow-up. In addition to congenital heart defects, many studies reported detection rates for other structural anomalies such as diaphragmatic hernia, skeletal dysplasia, abdominal wall defects, and central nervous system malformations. Follow-up periods extended from the immediate prenatal period to the neonatal stage. Characteristics of the included studies are summarized in Table 3 and Table 4.
Risk of bias assessment
Overall, most studies demonstrated low to moderate risk of bias in the domains of patient selection, index test, and flow and timing. Prospective cohort studies such as those by Hyett et al. (1999), Michailidis et al. (2001), and Volpe et al. (2011) generally reported clear inclusion criteria, standardized NT measurement protocols, and blinded outcome assessment, contributing to lower bias risk.
Conversely, some retrospective analyses (e.g., Tang et al., 2019; Kristensen et al., 2023) lacked detailed reporting on blinding or consistency in follow-up verification, raising concerns in the “reference standard” and “flow and timing” domains. Variability in NT cut-off values and measurement protocols across studies introduced applicability concerns, particularly in multicenter designs such as Jin et al. (2021) and Karadzov et al. (2019). Despite these limitations, the majority of included studies were considered methodologically sound enough to contribute meaningfully to the synthesis.
Diagnostic accuracy for congenital heart defects
Several more recent studies without detailed sensitivity/specificity reporting still contributed to the qualitative synthesis. For example, Zhang et al. (2023) reported a higher incidence of CHDs among fetuses with increased NT. Bottelli et al. (2023) focused on false-negative CHD cases, underscoring limitations of NT as a standalone screening tool, and Kristensen et al. (2023) examined mortality outcomes in CHD cases with increased NT. These findings reinforce that while NT can be a useful early marker, its sensitivity varies substantially and is often improved when combined with other ultrasound or biochemical markers.
Diagnostic accuracy for other major structural anomalies
Thirteen studies assessed NT’s performance for detecting non-cardiac major structural anomalies, including neural tube defects, abdominal wall defects, skeletal dysplasias, and genitourinary anomalies. Detection rates for these anomalies varied widely, with higher NT measurements generally associated with increased risk of major malformations, even in chromosomally normal fetuses.
Esteves et al. (2023) demonstrated that detailed ultrasound assessment in fetuses with increased NT improved detection of multiple structural anomalies beyond CHDs. Tang et al. (2022) found that NT remains a strong predictor for a range of anomalies in both low- and high-risk pregnancies. Karadzov et al. (2019) and Tang et al. (2019) showed improved screening performance when NT was combined with ductus venosus Doppler and tricuspid regurgitation assessment. Dulgheroff et al. (2019) and Dilek et al. (2023) reported that while first-trimester detection is valuable, second- and third-trimester scans still identify additional anomalies missed earlier.
Jin et al. (2021) and Zhang et al. (2023) both observed significant rates of extracardiac malformations among fetuses with isolated increased NT, supporting its use as a general structural anomaly risk marker. Abu-Rustum et al. (2010) and Timmerman et al. (2010) also included broader structural anomalies in their outcomes, reporting improved detection when NT measurement was part of a multi-parameter screening approach. Collectively, the evidence suggests that while NT has limitations as a standalone tool, its integration into comprehensive prenatal screening protocols enhances early detection of both cardiac and non-cardiac major anomalies (Hyett et al., 1999; Michailidis et al., 2001; Muller et al., 2007).
Discussion
The present systematic review synthesized evidence from 20 studies published between 1999 and 2023 evaluating the role of nuchal translucency (NT) measurement in detecting congenital heart defects (CHDs) and other major structural anomalies across both low- and high-risk pregnancies. Overall, NT enlargement was consistently associated with an increased risk of structural anomalies, although diagnostic accuracy varied widely across studies and conditions. While several investigations reported high specificity, sensitivity estimates for CHD detection ranged from 15% to 100%, reflecting heterogeneity in populations, NT cut-offs, and the use of adjunctive screening modalities. For non-cardiac anomalies, findings reinforced NT as a valuable but imperfect early screening marker, with optimal performance achieved when integrated into a comprehensive ultrasound and biochemical screening program.
In the study by Timmerman et al., the specificity of nuchal translucency (NT) in predicting congenital heart defects (CHD) was relatively low (62%) when compared to its sensitivity. Several factors may explain this finding. First, the study population was limited to euploid fetuses, since all cases with chromosomal abnormalities were excluded from the analysis. As a consequence, the number of fetuses with enlarged NT but without CHD remained relatively high, thereby inflating the false-positive rate and lowering specificity. This reflects the fact that enlarged NT is not only associated with structural cardiac anomalies, but also with chromosomal abnormalities and other genetic or developmental disorders, many of which were removed from the dataset.
For CHDs, pooled qualitative evidence indicated that NT has moderate diagnostic performance, with high specificity but variable sensitivity. This aligns with previous reviews which have similarly reported that increased NT is a significant but not definitive predictor of cardiac anomalies (11, 12). The variability in sensitivity among the included studies may be explained by differences in gestational age at the time of scanning, operator expertise, image resolution, and the spectrum of CHDs included in outcome definitions (13). Some defects, such as hypoplastic left heart syndrome or transposition of the great arteries, may be more likely to produce early cardiac hemodynamic changes and fluid accumulation in the nuchal region, leading to increased NT (14). In contrast, smaller septal defects or lesions with delayed hemodynamic impact may remain undetected in early gestation, contributing to false negatives (15). Physiologically, increased NT in CHDs may result from altered cardiac function, impaired venous return, or abnormal lymphatic development, which cause transient fluid accumulation in the fetal neck (16). However, because NT can also be elevated in chromosomally normal fetuses without CHDs, its predictive value improves substantially when combined with other first-trimester assessments such as ductus venosus Doppler, tricuspid regurgitation evaluation, or biochemical markers (17).
For other major structural anomalies, our synthesis supports prior findings that increased NT is associated with a wide range of extracardiac malformations, including skeletal dysplasias, diaphragmatic hernia, omphalocele, and renal anomalies (18). Several mechanisms may account for this association. For example, in skeletal dysplasias, delayed ossification and abnormal connective tissue development can impair lymphatic drainage, leading to NT fluid accumulation (19, 20). In abdominal wall defects, increased NT may reflect generalized fetal edema due to disrupted venous return (21). Furthermore, some anomalies may share underlying developmental pathways with cardiac defects, explaining their co-occurrence in certain cases (22). The reviewed studies also indicated that some extracardiac anomalies manifest later in gestation, which explains why NT alone cannot detect all cases and why second- and third-trimester follow-up is critical (23, 24). The literature emphasizes that while NT measurement is not a comprehensive anomaly screen, it serves as an early warning sign prompting targeted fetal echocardiography and detailed anatomical assessment (25). The mechanism linking increased NT to extracardiac anomalies remains multifactorial, involving genetic factors, structural malformations affecting lymphatic or venous flow, and early embryonic developmental disturbances (26, 27).
Despite the strengths of including a large number of studies spanning diverse populations and structural anomaly types, this review has several limitations. First, heterogeneity in NT cut-off values, gestational age at assessment, ultrasound protocols, and operator expertise complicates direct comparison of diagnostic performance across studies. Second, several included studies were retrospective or conducted in tertiary referral centers, potentially inflating diagnostic accuracy due to higher anomaly prevalence compared with the general population. Third, the absence of uniform follow-up protocols in some reports raises the possibility of underestimating false negatives, particularly for anomalies that manifest later in pregnancy. Fourth, publication bias cannot be excluded, as studies with negative or inconclusive findings may be underreported. Finally, although we incorporated both cardiac and extracardiac outcomes, the variability in how anomalies were classified and confirmed across studies limits the ability to derive pooled quantitative estimates.
Conclusion
This systematic review demonstrates that increased NT measurement is a valuable early screening tool for the detection of congenital heart defects and other major structural anomalies, though its diagnostic accuracy varies across anomaly types and study settings. NT shows particularly high specificity for cardiac anomalies, supporting its role as an effective triage marker for targeted fetal echocardiography and detailed anomaly scanning. For extracardiac structural anomalies, NT measurement can provide important early clues, but its performance is more variable and often requires integration with additional sonographic or biochemical markers.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors did not receive funding for this study.
Acknowledgements
The authors would like to express sincere appreciation to the staff who provided excellent technical support and assistance during the study.
Authors’ contributions
Dr. Elizabet Catherine Jusuf, M.Kes, Sp.OG(K) contributed to critical revision of the manuscript for important intellectual content. Dr. Karloman Agusto Paipinan contributed substantially to the concept, design, data analyses, interpretation of the data, and completion of the study and manuscript. All authors have read and approved the final manuscript.
Data availability statement
The data that support the findings of this study are available on request from Dr. dr. Elizabet Catherine Jusuf, M.Kes, Sp.OG(K) as corresponding author. The data are not publicly available due to privacy or ethical restrictions.
References
- Xu J, Li Q, Deng L, Xiong J, Cheng Z, Ye C. Global, regional, and national epidemiology of congenital heart disease in children from 1990 to 2021. Front Cardiovasc Med. 2025;12:1522644. doi: 10.3389/fcvm.2025.1522644.
- Colaco SM, Karande T, Bobhate PR, Jiyani R, Rao SG, Kulkarni S. Neonates with critical congenital heart defects: impact of fetal diagnosis on immediate and short-term outcomes. Ann Pediatr Cardiol. 2017;10(2):126–30. doi: 10.4103/apc.APC_125_16.
- Rayburn WF, Jolley JA, Simpson LL. Advances in ultrasound imaging for congenital malformations during early gestation. Birth Defects Res A Clin Mol Teratol. 2015;103(4):260–7. doi: 10.1002/bdra.23353.
- Barati M, Zargar M, Masihi S, Taherpour S. Evaluation of nuchal translucency measurement in first trimester pregnancy. Int J Fertil Steril. 2011; 5(1):35–8.
- Sofia-Gonçalves A, Guedes-Martins L. Nuchal translucency and congenital heart defects. Curr Cardiol Rev. 2024;20(2):1–13. doi:10.2174/ 1573403X20666240306100921.
- Li Y, Du J, Deng S, Liu B, Jing X, Yan Y, et al. The molecular mechanisms of cardiac development and related diseases. Signal Transduct Target Ther. 2024;9(1):368. doi: 10.1038/s41392-024-01768-x.
- Salman Guraya S. The associations of nuchal translucency and fetal abnormalities: significance and implications. J Clin Diagn Res. 2013; 7(5):936–41.
- Heazell AE, Hayes DJ, Whitworth M, Takwoingi Y, Bayliss SE, Davenport C. Biochemical tests of placental function versus ultrasound assessment of fetal size for stillbirth and small-for-gestational-age infants. Cochrane Database Syst Rev. 2019; 5(5):CD012245. doi:10.1002/ 14651858.CD012245.pub2.
- Frisova V. Prenatal screening for chromosomal defects. Prenat Diagn Rev. 2025;6(2):15.
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021; 372: n71. doi:10.1136/bmj.n71.
- Kristensen R, Omann C, Gaynor JW, Rode L, Ekelund CK, Hjortdal VE. Increased nuchal translucency in children with congenital heart defects and normal karyotype—is there a correlation with mortality? Front Pediatr. 2023; 11:1104179. doi:10.3389/fped.2023.1104179.
- Mogra R, Alabbad N, Hyett J. Increased nuchal translucency and congenital heart disease. Early Hum Dev. 2012; 88(5):261–7. doi: 10.1016/j.earlhumdev.2012.03.001.
- Ginsberg GM, Drukker L, Pollak U, Brezis M. Cost-utility analysis of prenatal diagnosis of congenital cardiac diseases using deep learning. Cost Eff Resour Alloc. 2024;22(1):44. doi:10.1186/s12962-024-00444-z.
- Orvos H, Wayda K, Kozinszky Z, Katona M, Pál A, Szabó J. Increased nuchal translucency and congenital heart defects in euploid fetuses: the Szeged experience. Eur J Obstet Gynecol Reprod Biol. 2002;101(2):124–8.
- Harris IS. Management of pregnancy in patients with congenital heart disease. Prog Cardiovasc Dis. 2011; 53(4):305–11. doi:10.1016/ j.pcad.2010.08.001.
- Alsaied T, Rathod RH, Aboulhosn JA, Budts W, Anderson JB, Baumgartner H, et al. Reaching consensus for unified medical language in Fontan care. ESC Heart Fail. 2021;8(5):3894–905. doi:10.1002/ehf2.13294.
- Karadzov Orlić N, Egić A, Damnj anović-Pažin B, Lukić R, Joksić I, Miković Z. Screening performance of congenital heart defects in first trimester using simple cardiac scan, nuchal translucency, abnormal ductus venosus blood flow and tricuspid regurgitation. Congenit Heart Dis. 2019;14(6):1094–101. doi:10.1111/ chd.12756.
- Al Namat D, Roșca RA, Al Namat R, Hanganu E, Ivan A, Hînganu D, et al. Omphalocele and associated anomalies: exploring pulmonary development and genetic correlations—a literature review. Diagnostics (Basel). 2025;15(6):675. doi:10.3390/diagnostics15060675.
- Sonek J. First trimester ultrasonography in screening and detection of fetal anomalies. Am J Med Genet C Semin Med Genet. 2007; 145C(1):45–61. doi:10.1002/ajmg.c.30120.
- Lee YB, Kim MJ, Kim MH. Robust border enhancement and detection for measurement of fetal nuchal translucency in ultrasound images. Med Biol Eng Comput. 2007;45(11):1143–52. doi:10.1007/s11517-007-0225-7.
- Abassi Z, Khoury EE, Karram T, Aronson D. Edema formation in congestive heart failure and the underlying mechanisms. Front Cardiovasc Med. 2022;9:933215. doi:10.3389/ fcvm.2022.933215.
- Zubrzycki M, Schramm R, Costard-Jäckle A, Grohmann J, Gummert JF, Zubrzycka M. Cardiac development and factors influencing the development of congenital heart defects (CHDs): part I. Int J Mol Sci. 2024; 25(13):7051. doi: 10.3390/ijms25137051.
- Sadlecki P, Walentowicz-Sadlecka M. Prenatal diagnosis of fetal defects and its implications on the delivery mode. Open Med (Warsaw). 2023; 18(1):20230704. doi:10.1515/med-2023-0204.
- Li YF, Hua YM, Fang J, Wang C, Qiao LN, Wan CM, et al. Performance of different scan protocols of fetal echocardiography in the diagnosis of fetal congenital heart disease: a systematic review and meta-analysis. PLoS One. 2013; 8(6): e65034. doi:10.1371/journal.pone.0065034.
- Sairam S, Carvalho JS. Early fetal echocardiography and anomaly scan in fetuses with increased nuchal translucency. Early Hum Dev. 2012; 88(5): 269–72. doi:10.1016/j.earlhumdev.2012.03.001.
- Humbert M, Kovacs G, Hoeper MM, Badagliacca R, Berger RMF, Brida M, et al. 2022 ESC/ERS guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Heart J. 2022; 43(38): 3618–731. doi:10.1093/eurheartj/ehac237.
- Zhang HJ, Wang S, Feng CL, Zhao HY, Zhang WW, Sun Y, et al. Chromosomal abnormalities and structural defects in fetuses with increased nuchal translucency at a Chinese tertiary medical center. Front Med (Lausanne). 2023; 10: 1123456. doi:10.3389/fmed.2023.1123456.
- Ximenes RS, Bravo-Valenzuela NJ, Pares DBS, Araujo EJ Jr. The use of cardiac ultrasound imaging in first-trimester prenatal diagnosis of congenital heart diseases. J Clin Ultrasound. 2023;51(2):225–39. doi:10.1002/jcu.23123.
- Esteves KM, Tugarinov N, Lechmann G, Habib PA, Cagliyan E, Goetzinger KR, et al. The value of detailed first-trimester ultrasound in the era of noninvasive prenatal testing. Am J Obstet Gynecol. 2023; 229(3): 276–84. doi:10.1016/ j.ajog.2023.04.012.
- Dilek TUK, Oktay A, Aygun EG, Ünsal G, Pata Ö. Evaluation of fetal heart in the first and second trimester: results and limitations. Niger J Clin Pract. 2023; 26(6): 787–94. doi:10.4103/njcp.njcp_1_23.
- Bottelli L, Franzè V, Tuo G, Buffelli F, Paladini D. Prenatal detection of congenital heart disease at 12–13 gestational weeks: detailed analysis of false-negative cases. Ultrasound Obstet Gynecol. 2023;61(5):577–86. doi:10.1002/uog.26094.
- Paija S, Shah J, Chudasama N, Vora H. Nuchal translucency as an indispensable screening tool for predicting congenital heart diseases. Eur J Mol Clin Med. 2022;9(7):2537–43.
- Jin H, Wang J, Zhang G, Jiao H, Zhu J, Li Z, et al. A Chinese multicenter retrospective study of isolated increased nuchal translucency associated chromosome anomaly and prenatal diagnostic suggestions. Sci Rep. 2021;11(1):5596. doi: 10.1038/s41598-021-85108-6.
- Dulgheroff FF, Peixoto AB, Petrini CG, Caldas T, Ramos DR, Magalhaes FO, et al. Fetal structural anomalies diagnosed during the first, second and third trimesters of pregnancy using ultrasonography: a retrospective cohort study. Sao Paulo Med J. 2019; 137(5):391–400. doi: 10.1590/1516-3180.2019.0210130919.
- Tang Y, Yang TZ, Zhu Q, Luo H. The value of increasing nuchal translucency in diagnosis of congenital heart disease in fetus. Chin J Evid Based Med. 2015;15(4):373–6.
- Borrell A, Grande M, Bennasar M, Borobio V, Jimenez JM, Stergiotou I, et al. First-trimester detection of major cardiac defects with the use of ductus venosus blood flow. Ultrasound Obstet Gynecol. 2013;42(1):51–7. doi:10.1002/uog.12349.
- Eleftheriades M, Tsapakis E, Sotiriadis A, Manolakos E, Hassiakos D, Botsis D. Detection of congenital heart defects throughout pregnancy; impact of first trimester ultrasound screening for cardiac abnormalities. J Matern Fetal Neonatal Med. 2012;25(12):2546–50. doi:10.3109/14767058.2012.699120.
- Volpe P, Ubaldo P, Volpe N, Campobasso G, De Robertis V, Tempesta A, et al. Fetal cardiac evaluation at 11–14 weeks by experienced obstetricians in a low-risk population. Prenat Diagn. 2011;31(11):1054–61. doi:10.1002/pd.2840.
- Pereira S, Ganapathy R, Syngelaki A, Maiz N, Nicolaides KH. Contribution of fetal tricuspid regurgitation in first-trimester screening for major cardiac defects. Obstet Gynecol. 2011; 117(6): 1384–91. doi:10.1097/AOG.0b013e31821e8af2.
- Timmerman E, Clur SA, Pajkrt E, Bilardo CM. First-trimester measurement of the ductus venosus pulsatility index and the prediction of congenital heart defects. Ultrasound Obstet Gynecol. 2010;36(6):668–75. doi:10.1002/uog.7744.
- Abu-Rustum RS, Daou L, Abu-Rustum SE. Role of first-trimester sonography in the diagnosis of aneuploidy and structural fetal anomalies. J Ultrasound Med. 2010;29(10):1445–52.
- Müller MA, Clur SA, Timmerman E, Bilardo CM. Nuchal translucency measurement and congenital heart defects: modest association in low-risk pregnancies. Prenat Diagn. 2007; 27(2):164–9. doi:10.1002/pd.1659.
- Lombardi CM, Bellotti M, Fesslova V, Cappellini A. Fetal echocardiography at the time of the nuchal translucency scan. Ultrasound Obstet Gynecol. 2007;29(3):249–57. doi:10.1002/uog.4038.
- Michailidis GD, Economides DL. Nuchal translucency measurement and pregnancy outcome in karyotypically normal fetuses. Ultrasound Obstet Gynecol. 2001;17(2):102–5. doi:10.1046/j.0960-7692.2001.00321.x.
- Hyett J, Perdu M, Sharland G, Snijders R, Nicolaides KH. Using fetal nuchal translucency to screen for major congenital cardiac defects at 10–14 weeks of gestation: population based cohort study. BMJ. 1999;318(7176):81–5. doi: 10.1136/bmj.318.7176.81.